This post includes experimental procedures involving lithium-ion cells, battery packs, and embedded electronic components. All experiments described here were performed in a controlled environment at Sensloop Lab.
For safe experimentation, we recommend the following:
- Wear proper insulation gear and protective equipment (gloves, safety goggles, etc.).
- Check voltage and perform insulation measures before starting any work.
- Handle embedded circuits, sensors, and motor drivers with care to avoid short circuits or electrical damage.
- High-voltage battery pack or high-current embedded testing should be conducted only by trained personnel.
💡 Research Overview
Lithium-ion battery thermal runaway is the primary failure mechanism behind most EV battery fires and large-scale ESS incidents.
Although such events are often described as “explosions,” the internal process is more accurately defined as a self-accelerating exothermic reaction sequence. Within seconds, heat accumulation triggers electrolyte decomposition, oxygen release from cathode materials (NCM/NCA), separator failure, and internal short circuits.
This article explains:
- How thermal runaway begins at the cell level
- Why it propagates across an EV battery pack
- Why re-ignition frequently occurs after suppression
🔥 1. What Is Thermal Runaway?
Thermal runaway occurs when:
The rate of internal heat generation exceeds the rate of heat dissipation.
In lithium-ion cells (18650, 21700, pouch, prismatic), heat may originate from:
- Overcharge
- Over-discharge
- Internal short circuit
- Mechanical damage
- External heating
When heat generation surpasses cooling capacity, the internal temperature rises exponentially. Once critical thresholds are reached (typically above 120–150°C), decomposition reactions accelerate uncontrollably.
This leads to a cascade of exothermic reactions, not a single event.
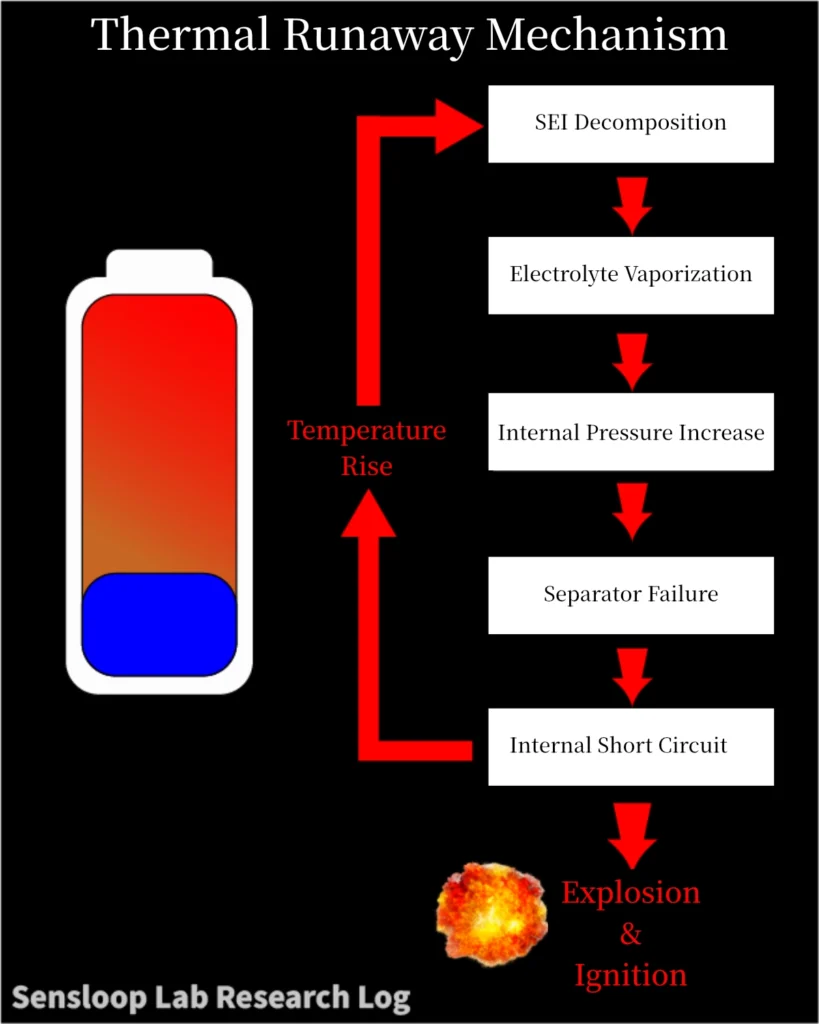
⚙️ 2. The HTR Loop: Heat–Temperature–Reaction Cycle
Thermal runaway progresses through a reinforcing feedback cycle known as the HTR loop (Heat–Temperature–Reaction).
| Stage | Key Event | Technical Mechanism |
|---|---|---|
| ① Heat Initiation | SEI Decomposition | The Solid Electrolyte Interphase layer breaks down, releasing heat and gases. |
| ② Temperature Rise | Electrolyte Vaporization | Organic electrolyte decomposes and vents flammable gases (CO₂, HF, hydrocarbons). |
| ③ Reaction Acceleration | Cathode/Anode Decomposition | Oxygen release from cathode material increases reaction intensity. Internal pressure rises rapidly. |
| ④ Catastrophic Escalation | Separator Failure & Internal Short Circuit | Separator melts (~130–170°C), electrodes contact directly → sudden electrical discharge. |
Each stage feeds the next:
Heat → Temperature Increase → Faster Chemical Reaction → More Heat
If uninterrupted, the cell enters a self-sustaining combustion state.

⚡ 3. Why Does One Cell Ignite an Entire EV Battery Pack?
EV and ESS battery packs consist of hundreds to thousands of tightly packed cells.
When a single cell undergoes thermal runaway:
- Heat transfers to neighboring cells.
- Flammable gases accumulate within confined pack structures.
- Adjacent cells reach decomposition thresholds.
This domino phenomenon is called:
Thermal Propagation
In EV battery packs, structural confinement and limited ventilation amplify pressure buildup. One failed 18650 or 21700 cell can initiate sequential failure across modules.
Thermal propagation is often responsible for large-scale vehicle fires.

🧊 4. Why Is Lithium-Ion Battery Fire Suppression So Difficult?
Unlike conventional fires, lithium-ion battery fires involve internal chemical reactions.
Even after visible flames are extinguished:
- Electrolyte may continue decomposing.
- Residual heat remains trapped.
- Internal short circuits may persist.
- Cathode materials may continue oxygen release.
This explains the high risk of:
Re-ignition hours after initial suppression
Multiple EV incidents (including Tesla Model S and Hyundai Kona EV reports) documented delayed re-ignition.
External cooling alone is often insufficient.

🧪 5. Sensloop Lab Technical Analysis Summary
| Condition | Before Runaway | During Runaway | Post-Suppression Risk |
|---|---|---|---|
| Temperature | 30–80°C | 150–800°C Rapid Surge | Elevated re-ignition probability |
| Internal Reaction | Stable charge/discharge | SEI breakdown + electrolyte decomposition | Internal short + residual gas pressure |
| Suppression Effectiveness | Surface cooling effective | Limited | Continuous cooling required |
Complete suppression requires:
Simultaneous heat removal and internal reaction termination.
Standard CO₂ or dry powder extinguishers do not address internal exothermic reactions.

🧠 6. Conclusion: Thermal Runaway Is a Time-Critical Failure Mechanism
Lithium-ion battery fires are multi-factor events involving:
- Electrical failure
- Chemical decomposition
- Mechanical separator collapse
- Oxygen release
Thermal runaway is fundamentally a race against time.
Early detection, rapid cooling, and prevention of propagation within EV battery packs are essential for effective fire mitigation.
Understanding this mechanism is critical for:
- EV battery safety engineering
- ESS fire prevention systems
- Advanced battery management systems (BMS)
🔗 Next Article
In the next post, we will examine:
How overcharge and over-discharge trigger lithium-ion battery thermal runaway.

